There is something almost absurdly audacious about the question scientists have been wrestling with for centuries: how did something alive come from absolutely nothing alive? You’re here, reading these words, built from trillions of cells, each one a tiny humming factory running on chemistry that stretches back nearly four billion years. And yet nobody told those first molecules what to do. They just… figured it out.
The origin of life is not just a scientific mystery. It is, honestly, the most profound detective story in all of human history. The evidence is ancient, the clues are hidden in rocks and chemistry and genetic code, and every time scientists think they’re close to a final answer, the story deepens. So buckle up, because this is one rabbit hole worth following all the way down.
A Planet You Would Barely Recognize: The Early Earth

Picture a world that looks nothing like the one you live in now. No trees, no oceans teeming with fish, no breathable air. The primordial soup represents the hypothetical set of conditions present on the Earth around 3.7 to 4.0 billion years ago. You would be standing on a surface battered by volcanic eruptions and pelted by asteroids, breathing an atmosphere with virtually no oxygen.
According to early scientific thinking, the primitive Earth’s atmosphere was essentially reducing, with little or no oxygen, and ultraviolet rays from the Sun induced reactions on a mixture of water, carbon dioxide, and ammonia. Think of it like a chemistry lab that nobody designed, with no walls, no safety protocols, and absolutely no supervision. That, remarkably, is where the story of you begins.
The atmosphere of primitive Earth was reducing in nature, with large amounts of gases like methane, ammonia, water vapor, and very little oxygen. Various sources of energy, such as heat, lightning, and UV rays, were abundant on this primitive Earth. It was a wild, almost hostile world. Yet somehow, that hostility turned out to be exactly the right kind of creative pressure.
The Primordial Soup: Life’s First Kitchen
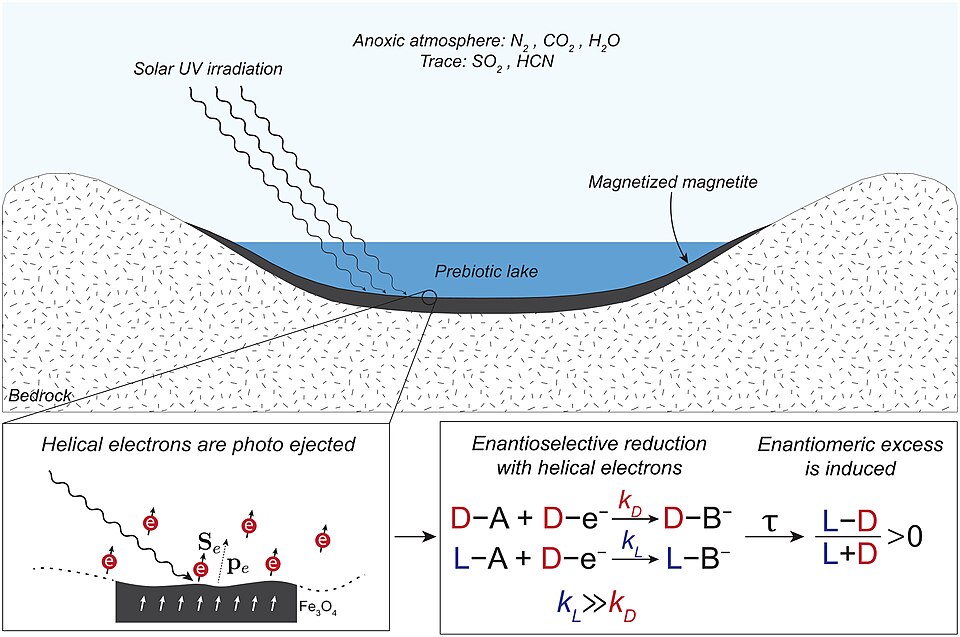
Imagine a giant, planet-wide broth that nobody stirred intentionally, yet somehow produced the most complex recipe in the known universe. According to theory, organic compounds essential for life forms were synthesized in the primitive Earth under prebiotic conditions. The mixture of inorganic and organic compounds with water on the primitive Earth became the prebiotic or primordial soup. There, life originated and the first forms of life were able to use the organic molecules to survive and reproduce.
According to the primordial soup theory, the first forms of life originated from primitive water bodies on Earth, which contained complex organic matter. The organic matter that led to the formation of life is believed to have formed from inorganic substances present in the reducing atmosphere of the early Earth. I think what makes this idea so compelling is just how ordinary the ingredients are. Carbon. Hydrogen. Nitrogen. Water. You probably have all of those in your kitchen right now, in slightly different forms.
The primordial soup did not encompass all the oceans of the primitive Earth. Instead, it applied to some specific regions, such as intertidal zones, ocean sediments, shallow ponds, freshwater lakes, lagoons, and glacial ponds. So this wasn’t one massive global event. It was more like thousands of small local experiments running simultaneously, and at least one of them eventually hit the jackpot.
The Miller-Urey Experiment: Science Recreates the Impossible
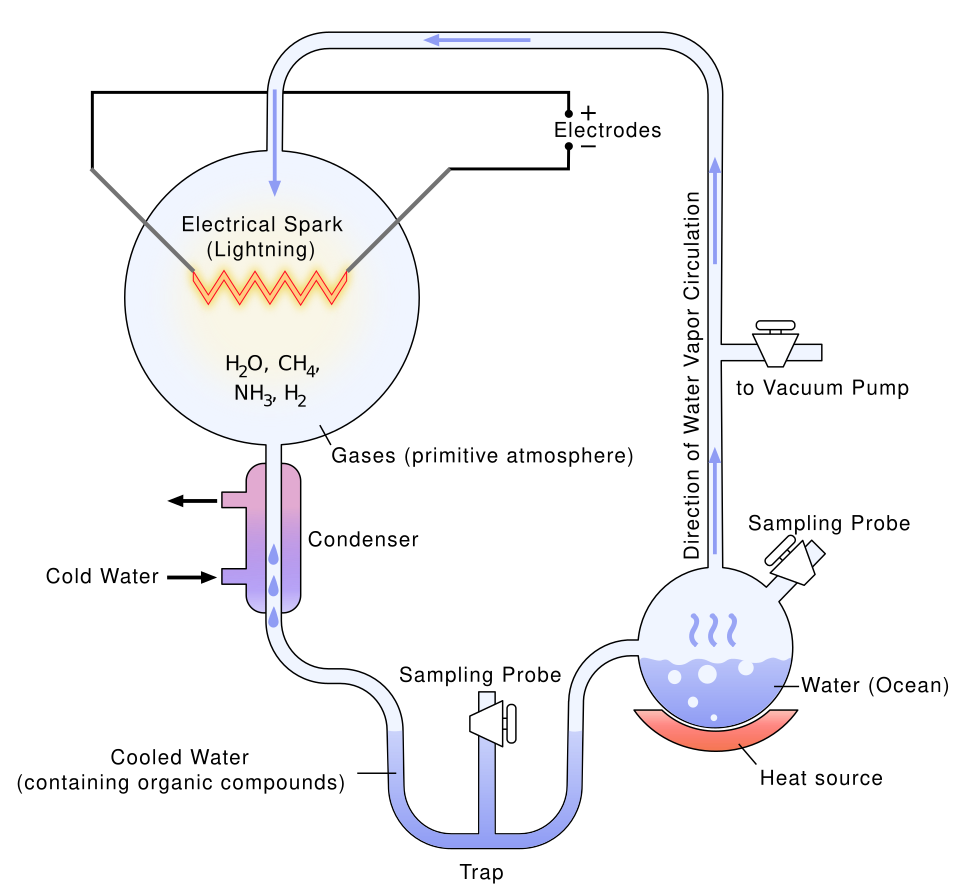
Here’s the thing: for a long time, the primordial soup was just an idea, a beautiful hypothesis with no proof. Then, in 1953, a young chemist named Stanley Miller changed everything. The Miller-Urey experiment was an experiment in chemical synthesis carried out in 1952 that simulated the conditions thought at the time to be present in the atmosphere of the early, prebiotic Earth. It is seen as one of the first successful experiments demonstrating the synthesis of organic compounds from inorganic constituents in an origin of life scenario.
The experiment documented the production of amino acids and other organic molecules, thereby demonstrating that chemical evolution, meaning the formation of complex chemicals from simple ones, is possible. In one afternoon in a lab in Chicago, the boundary between chemistry and biology started to blur. The Miller-Urey experiment sparked research on how simple organic molecules might polymerize into more complex molecules, a process that may have produced the first living cells.
More recently, scientists revisited the experiment with a surprising new twist. Research published in Science Advances builds upon the landmark 1953 Miller-Urey experiment. Electrical exchanges between oppositely charged water droplets can result in the production of amino acids. This process could have been more frequent than lightning on ancient Earth, creating abundant building blocks for life. So you don’t even necessarily need a dramatic lightning bolt. Tiny sparks in water droplets may have done much of the work, quietly and constantly, across the early planet.
Deep-Sea Hydrothermal Vents: Life’s Furnace at the Bottom of the Ocean

Not everyone is convinced the primordial soup told the whole story. There’s a rival theory, and it takes place in one of the most dramatic environments on Earth: the crushing darkness of the deep ocean floor. Since their discovery, hydrothermal vents have become the most popular theory among scientists for explaining the origins of life on Earth. The idea is that life didn’t start with sunlight and lightning. It started in total darkness, fuelled by chemistry bubbling up from inside the planet itself.
The environmental conditions in porous hydrothermal vents, where heated, mineral-laden seawater spews from cracks in the ocean crust, created a gradient in positively charged protons that served as a “battery” to fuel the creation of organic molecules and proto-cells. Think of it as a natural power outlet, one that never needed to be plugged in. That battery powered the chemical transformation of carbon dioxide and hydrogen into simple carbon-based molecules such as amino acids or proteins. Eventually that gradient drove the creation of cellular membranes, complicated proteins, and ribonucleic acid.
The study suggests hydrothermal vents, which probably littered the seafloor soon after the first oceans formed about 4.2 billion years ago, were not only a source of phosphorus, essential for the origin and early evolution of life, but also produced trillions upon trillions of tiny, highly reactive clay particles. Phosphorus is critical for building DNA and RNA. The fact that vents were pumping it out in abundance is no small detail. It’s arguably one of the most important clues we’ve found in recent years.
The RNA World: Life’s First Software
One of the most fundamental puzzles in biology is a classic chicken-and-egg problem. One of the challenges in studying abiogenesis is that the system of reproduction and metabolism utilized by all extant life involves three distinct types of interdependent macromolecules: DNA, RNA, and proteins, none of which can function and reproduce without the others. This suggests that life could not have arisen in its current form, which has led researchers to hypothesize mechanisms whereby the current system might have arisen from a simpler precursor system.
Enter RNA, the middle child of genetic molecules, and possibly the hero of this whole story. One view is that an RNA world existed on Earth before modern cells arose. According to this hypothesis, RNA stored both genetic information and catalyzed the chemical reactions in primitive cells. Only later in evolutionary time did DNA take over as the genetic material and proteins become the major catalyst and structural component of cells. RNA, in other words, wore all the hats before DNA and proteins arrived to specialize.
Chemists have shown how two of biology’s most fundamental ingredients, RNA and amino acids, could have spontaneously joined together at the origin of life four billion years ago. That’s a stunning finding. It’s like discovering that the pen, the ink, and the paper all assembled themselves. These new findings strengthen the RNA world hypothesis that describes how the first life on Earth could have used RNA instead of DNA. It’s hard to say for sure whether RNA came first in all cases, but the evidence is mounting in a way that’s hard to ignore.
The Oldest Fossils: Life’s First Timestamps

If you want to know when life first showed up, you look at rocks. And the rocks have told us something genuinely astonishing. Perhaps the earliest known signs of life have been found in Quebec, where features such as haematite tubes suggest that filamentous microbes lived around hydrothermal vents at least 3,770 million years ago. That is almost four billion years ago. The universe itself is only around 13.8 billion years old, for context.
Lead researchers analyzed jasper rock samples, finding microfossils of filaments and tubes filled with hematite, the mineral form of ferric oxide, similar to the remains of modern-day microbes living around hydrothermal vents. The presence of carbonate and carbonaceous minerals provided supporting signs of oxidation and biological activity. These are not flashy dinosaur bones. They’re tiny chemical whispers in ancient stone. Yet each one tells us that life did not take long to get started once Earth had cooled enough to allow it.
An earlier report of ancient life found in stromatolites from Greenland dating back 3.7 billion years was published during the summer of 2016, and the discovery of an ancient microbial ecosystem unearthed in the Pilbara of Western Australia that dated back 3.48 billion years was published in Astrobiology. Multiple rock formations on multiple continents are all pointing to the same jaw-dropping conclusion: life showed up on Earth fast, and it showed up everywhere it could.
What This Means for Life Beyond Earth

Here is where the story stops being purely historical and becomes genuinely thrilling for the present day. Discovering life elsewhere in the universe or creating it in the laboratory would constitute a profound scientific breakthrough, reshaping our understanding of biology, evolution, and the uniqueness of life on Earth. In the search for extraterrestrial life, two promising avenues are actively being pursued. Scientists are no longer just asking how life began here. They’re asking whether the same thing happened somewhere else.
The first avenue involves exploring our solar system, particularly environments such as Mars, Europa, and Enceladus, where liquid water and geochemical activity can support microbial ecosystems. The second focuses on detecting biosignatures from exoplanets, using next-generation telescopes to identify atmospheric gases or surface features that may indicate biological activity. Think about that for a moment. The same vents, the same chemistry, the same basic recipe that may have created life here could theoretically be at work right now beneath the icy crust of one of Saturn’s moons.
Breakthrough research aligns with the hypothesis that stable fatty acid membranes could have originated in alkaline hydrothermal vents, potentially progressing into living cells. The authors speculated that similar chemical reactions might unfold in the subsurface oceans of icy moons, which are thought to have hydrothermal vents similar to terrestrial ones. If life followed the hydrothermal vent pathway on Earth, there is no reason, scientifically speaking, why it couldn’t follow the same pathway on Enceladus or Europa. The universe, it turns out, may be far less lonely than we once imagined.
Conclusion: A Story That’s Far From Over

is, at its heart, a story about chemistry getting ambitious. From a barren, volcanic rock with a toxic atmosphere, through pools of primordial soup and the darkness of the deep ocean floor, through self-replicating molecules and the first trembling proto-cells, life found a way. Not because it was destined to, not because anything guided it, but because the conditions were right and the chemistry was patient.
The origin of life is perhaps the most interdisciplinary question in all of science. It demands biologists, chemists, geologists, astronomers, and physicists all sitting at the same table, which is exactly what is happening more and more in research institutions around the world today. Each new discovery adds a piece to the puzzle of the origin of life. None of us has seen the completed picture yet.
What fills me with genuine wonder is that those first molecules had no idea what they were starting. Four billion years later, their distant descendants are reading articles about them, wondering how it all began. The story isn’t over. In many ways, we’ve barely opened the first page. So, what do you think: does life feel more miraculous or more inevitable now that you know the story? Share your thoughts in the comments.




