You probably grew up with a simple picture of mammal evolution: tiny shrew‑like creatures scurrying under dinosaur feet, slowly turning into the cats, whales, and humans you know today. But the latest genetic and fossil evidence is quietly blowing up that tidy story. As more genomes are sequenced and new fossils are scanned in stunning detail, you start to see a much stranger, messier, and more fascinating origin story for mammals.
Instead of a straight line from reptile‑like ancestors to modern mammals, you’re looking at a tangled web of lineages that experimented with different jaws, ears, metabolisms, and ways to reproduce. Genetic data is now revealing surprising connections between groups you thought were far apart, and unexpected reversals where traits seem to evolve, disappear, and then re‑emerge in different forms. When you put the genetic and fossil clues together, early mammal evolution stops looking like a slow climb and starts to feel more like a thriller.
How Genomes Are Rewriting the Mammal Family Tree

If you still picture evolutionary trees as simple branching diagrams from school textbooks, you’re already behind what modern genomics is showing you. Over the last two decades, huge DNA datasets from dozens, then hundreds, of mammal species have forced researchers to redraw deep branches of the mammal family tree. With large genomic matrices, you can now test whether long‑standing fossil‑based groupings really hold up once you compare thousands of genes side by side. ([pubmed.ncbi.nlm.nih.gov](https://pubmed.ncbi.nlm.nih.gov/39173812/?utm_source=openai))
What you see in those genetic trees is not just finer detail, but some genuine shocks. Relationships that once seemed obvious based on bones and teeth sometimes fall apart once genomes are added, while previously puzzling links suddenly make sense. Recent phylogenomic work, including studies of placental mammals and carnivore relatives, has shown that a small subset of carefully chosen genes can sometimes resolve deep conflicts better than simply piling on more data. That means you have to think less about “more DNA equals truth” and more about which parts of the genome hold the most reliable signal about ancient splits. ([pubmed.ncbi.nlm.nih.gov](https://pubmed.ncbi.nlm.nih.gov/39173812/?utm_source=openai))
Jaw Joints and Middle Ears: A Patchwork, Not a Straight Line

You were probably told that one of the defining steps toward true mammals was the transformation of jaw bones into middle ear bones: a neat, one‑directional transition from a reptile‑style jaw joint to a mammalian one. Fresh fossil and imaging data are now telling you that this story is far more patchy and experimental than anyone expected. Tiny fossils from Brazil, for example, reveal that some early mammal relatives independently evolved jaw joint configurations that resemble the mammalian condition without being true mammals at all. ([nature.com](https://www.nature.com/articles/s41586-024-07971-3?utm_source=openai))
When you combine these fossils with high‑resolution scans of Jurassic mammaliaforms from China and elsewhere, you see multiple parallel experiments in how to separate chewing from hearing. The familiar mammalian middle ear did not flare into existence in one single ancestor; instead, it emerged through a mosaic of partial detachments and shifting connections across different lineages. That means when you look at your own ear bones today, you are seeing the final winner of a long series of evolutionary prototypes, many of which left only fragmentary traces in rock. ([amnh.org](https://www.amnh.org/explore/news-blogs/fossil-discoveries-mammal-evolution?utm_source=openai))
Teeth, Diet, and the Hidden Diversity of Early Mammaliaforms

If you want to understand how early mammals really lived, you have to stare at their teeth. Recent work on Jurassic mammaliaforms has revealed a surprising burst of dental innovation very early in mammal history, with some groups evolving specialized, multi‑cusped teeth that hint at a wide range of diets. In newly described shuotheriids and other Jurassic lineages, you can trace elaborate patterns of cusps and basins that suggest insect‑eating, omnivory, and perhaps even more complex feeding strategies than you might expect from “small nighttime insect munchers.” ([nature.com](https://www.nature.com/articles/s41586-024-07258-7?utm_source=openai))
When genetic data is added to this dental story, you start to see how repeated shifts in diet and jaw function may have been linked to deeper genomic changes in developmental and metabolic genes. It is not just that teeth diversified once and then stabilized; different mammal lineages appear to have explored overlapping “solutions” to similar ecological challenges, sometimes converging on comparable tooth shapes from different starting points. For you, that means the fossil jaws you see in museum cases are less a static ladder and more a record of competing evolutionary experiments that modern genomic tools are only now letting you connect.
Life Histories in Slow Motion: What Teeth Say About Growth and Aging
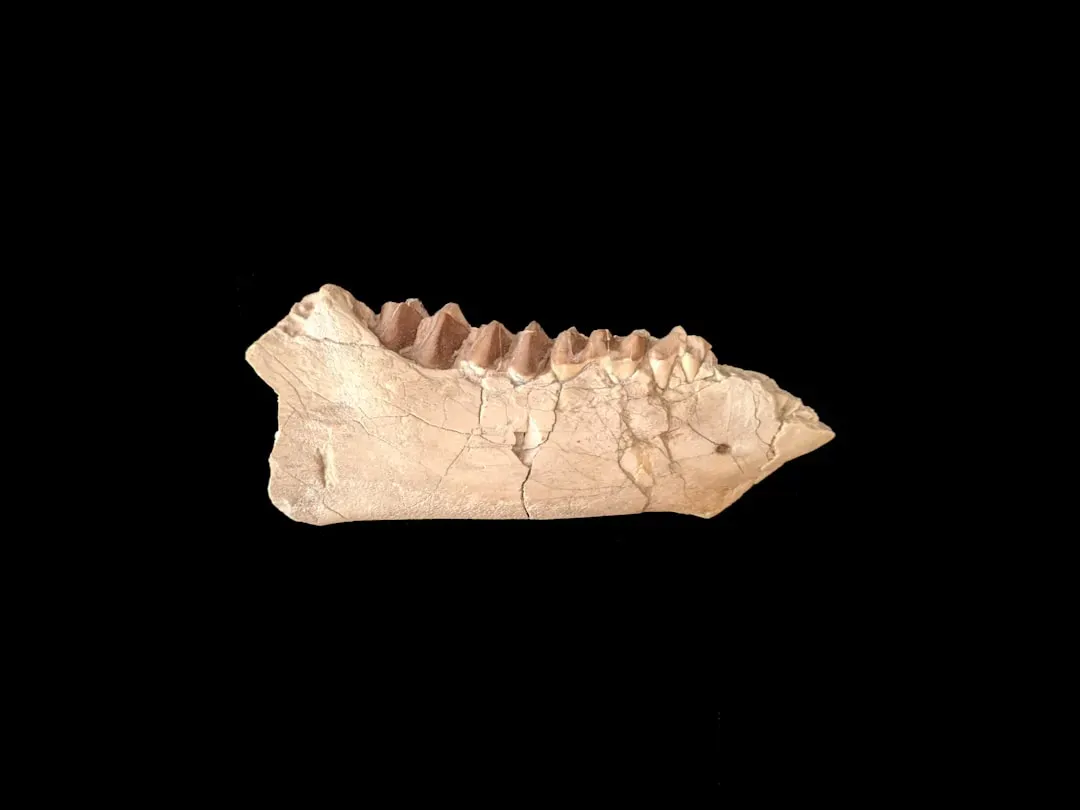
You might assume that early mammals already lived fast and died young, like many small mammals today. But growth rings in fossil teeth are now telling you a different story. By counting incremental layers in the roots of Jurassic mammaliaforms, researchers have shown that these early species often grew more slowly, lived longer, and reached sexual maturity later than mammals of similar size alive today. Even early crown mammals, closer to the modern condition, show growth patterns that sit somewhere between reptile‑like and fully mammalian life histories. ([sciencedaily.com](https://www.sciencedaily.com/releases/2024/08/240808115301.htm?utm_source=openai))
From your perspective, this means the “mammal way of life” did not appear overnight. High metabolic rates, extended parental care, and rapid early growth seem to have emerged stepwise, with different traits assembling at different times. Genetic evidence, especially from studies of metabolism‑related genes and regulatory networks, supports the idea that shifts in growth and energy use unfolded gradually, not in one sudden leap. So when you think about early mammals hiding under dinosaur shadows, you should picture creatures whose pace of life was still in transition, carrying a mix of ancestral and new traits in both their bodies and genomes. ([pubmed.ncbi.nlm.nih.gov](https://pubmed.ncbi.nlm.nih.gov/39110800/?utm_source=openai))
Eggs, Placentas, and the Surprising History of Mammalian Reproduction

It is easy to take live birth and placentas for granted, but the deeper you look into mammal origins, the stranger reproductive evolution becomes. At the fossil level, recent work on synapsid ancestors has strengthened the case that the earliest mammal precursors laid eggs, aligning them more closely with modern monotremes than you might expect. That means your own lineage ultimately traces back through creatures that combined some mammal‑like features with a reproductive strategy you would now associate with reptiles and platypuses. ([sciencedaily.com](https://www.sciencedaily.com/releases/2026/04/260414075642.htm?utm_source=openai))
On the genetic side, large comparative genomic and transcriptomic datasets are revealing unexpected links between very different kinds of mammals through their placental and germline genes. Studies of placental evolution suggest that complex, invasive placentas arose earlier and in more lineages than older models proposed, with some groups later reverting to less invasive forms. Research on spermatogenesis in marsupials and placentals has also highlighted novel gene expression patterns that appear to have been fine‑tuned in the placental lineage after its split from marsupials roughly one hundred and sixty million years ago. When you see these patterns together, you realize mammalian reproduction is not a one‑way march from eggs to live birth, but a branching set of paths with surprising reversals and refinements. ([pubmed.ncbi.nlm.nih.gov](https://pubmed.ncbi.nlm.nih.gov/39173812/?utm_source=openai))
Brains, Senses, and the Slow Rise of “Mammal Minds”

You might be tempted to imagine that once mammals appeared, big brains and keen senses followed quickly, but the fossil record argues for a slower build. Recent syntheses of synapsid brain evolution, based on internal cranial casts and inner ear anatomy, show that key mammalian sensory features such as enhanced hearing and tactile sensitivity evolved in stages. Early mammaliaforms already show inner ear structures trending toward the highly sensitive organs you rely on for balance and hearing today, yet many aspects of brain organization still look intermediate between reptile‑like ancestors and modern mammals. ([cambridge.org](https://www.cambridge.org/core/services/aop-cambridge-core/content/view/500B0F6CD3E934B1B9D29D5EBF2B1C83/S0022336025101212a.pdf/150_years_of_synapsid_paleoneurology_the_origins_of_the_mammalian_brain_behavior_sense_organs_and_physiology.pdf?utm_source=openai))
Genetic data adds another layer by letting you trace when certain neural and sensory gene families expanded or changed their regulation. While the details are still being worked out, current evidence suggests that some molecular building blocks for advanced mammalian behavior evolved before fully modern brains did, setting the stage for later bursts of innovation. When you think about that, it feels less like a light switch being flipped and more like a dimmer slowly turning up, with early mammals gradually entering a richer sensory and behavioral world long before primates or humans came on the scene.
Deep‑Time DNA and the Power – and Limits – of Genetic Clues

You are living at a moment when genetic tools are powerful enough to reach further back into time than anyone thought possible a generation ago. Ancient DNA studies have already transformed how you understand relatively recent mammal history, such as the rise and fall of different mammoth lineages. Even though DNA from the earliest mammals themselves is far beyond reach, those Pleistocene genomes serve as a proof of concept: by combining genetic clocks, mutation patterns, and living diversity, you can push inferences much deeper into the Mesozoic. ([uu.diva-portal.org](https://uu.diva-portal.org/smash/get/diva2%3A1954704/FULLTEXT01.pdf?utm_source=openai))
At the same time, the latest mammal phylogenomics reminds you to be cautious. Different parts of the genome can tell conflicting stories, and biases such as base composition can mislead even the most sophisticated analyses if you are not careful about gene choice and model design. Some recent studies have shown that focusing on subsets of genes less prone to systematic error can actually clarify contentious deep branching patterns among placental mammals. For you, that means genetic evidence is not a magic oracle; it is a powerful but noisy signal that has to be constantly cross‑checked against anatomy, fossils, and basic evolutionary logic. ([pubmed.ncbi.nlm.nih.gov](https://pubmed.ncbi.nlm.nih.gov/39173812/?utm_source=openai))
What These Unexpected Links Mean for How You See Yourself

When you pull all of this together – jaws, ears, teeth, growth, reproduction, brains, and genomes – you start to see your own place in the mammal story very differently. You are not the product of a clean, stepwise progression, but the descendant of lineages that tried multiple versions of key traits before settling on the combinations that define modern mammals. Traits you think of as quintessentially mammalian, from middle ears to placentas and complex life histories, turn out to have messy, overlapping origins marked by convergence, reversals, and long periods of experimentation.
There is something oddly grounding about that realization. Instead of a heroic climb toward some predetermined endpoint, early mammal evolution looks more like a series of bets placed under intense ecological pressure, many of which failed while a few reshaped life on Earth. As genomic technologies advance and more fossils are scanned or discovered, you can expect even more surprises about who is related to whom and how key mammal traits really evolved. In the end, the most important shift might be in how you think about evolution itself: not as a simple ladder, but as a restless, creative process that keeps rewriting its own history – sometimes in ways you never saw coming. When you look in the mirror after that, do you feel just a bit more connected to that long, experimental past?



